 
In this case, the reaction is highly exothermic, and the drive towards a decrease in energy allows the reaction to occur. According to the drive towards higher entropy, the formation of water from hydrogen and oxygen is an unfavorable reaction. 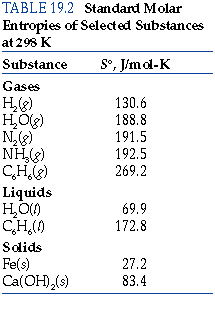
The entropy change for this reaction is highly negative because three gaseous molecules are being converted into two liquid molecules. This text is adapted from Openstax, Chemistry 2e, Chapter 16.2: The Second and Third Law of Thermodynamics. Download Table shows standard enthalpy of formation, standard entropy, standard molar heat capacity, and standard mclar volume of calcite, doiomite. The value for Δ S° rxn is negative, as expected for this phase transition (condensation).Īs a second example, consider the combustion of methanol, CH 3OH: 
The standard entropy changes for the reaction, Δ S° rxn is calculated using the standard molar entropies and stoichiometric coefficients. SubstanceĬonsider the condensation of water, in which 1 mole of gaseous H 2O changes into 1 mole of liquid H 2O. Where n p and n r represent the stoichiometric coefficients in the balanced equation of the products and reactants, respectively.įor example, Δ S° rxn for the following reaction at room temperatureĪ partial listing of standard entropies is provided in the table. 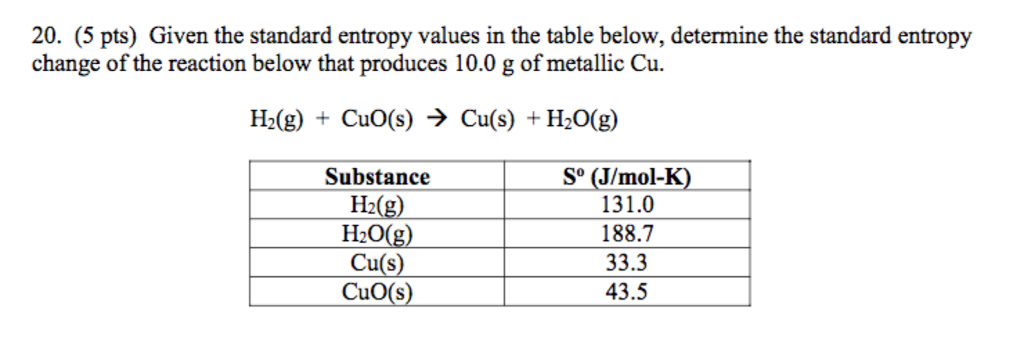
Therefore, entropy decreases as the reaction proceeds.Įntropy is a state function, so the standard entropy change for a chemical reaction (Δ S° rxn) can be calculated from the difference in standard entropy between the products and the reactants. Thus, in this reaction, the reactants are more disordered than the products. There are more moles of gas in the reactants, 4 moles of gas (with 1 mole of ethylene and 3 moles of oxygen) compared to the products (only 2 moles of carbon dioxide gas), while the other product is a liquid. Recall that gases are more disordered than liquids. The negative value indicates there is a decrease in entropy.Įven without calculating the exact entropy change, the decrease in entropy can be predicted by examining the reaction. The difference between the products and the reactants equals negative 268 J/K for the standard entropy change of the combustion of ethylene. The net entropy of the products equals 567.6 J/K, and the net entropy of the reactants is 835.4 J/K. Substituting the values for molar entropies of reactants and products from the reference table yields −. Note that, unlike standard enthalpies of formation of elements, which are zero, standard molar entropies of all substances are greater than zero at 298 K. Entropy of gas at standard conditions (1 bar) Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. The standard entropy change for the reaction equals the sum of 2 times the standard entropy of carbon dioxide gas and 2 times the standard entropy of water, minus the sum of the standard entropy of ethylene gas and 3 times the standard entropy of oxygen. So, like enthalpy changes, entropy changes can be from calculated reference tables of standard molar entropies.įor a reaction occurring under standard conditions, the associated entropy change is determined by the difference between the sum of the standard molar entropies of the products multiplied by their stoichiometric coefficients and the sum of the standard molar entropies of the reactants multiplied by their stoichiometric coefficients.Ĭonsider the combustion of ethylene under standard conditions, where 1 mole of ethylene gas reacts with 3 moles of oxygen gas to produce 2 moles of carbon dioxide gas and 2 moles of water. Enthalpy changes associated with a chemical reaction can be measured with a calorimeter, but the entropy change associated with a reaction cannot be directly measured.Įntropy is a state function, which means that the change in entropy depends solely on the initial and final states of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
